
Back هاليد الأسيل Arabic Acylhalid Danish Säurehalogenide German Haluro de ácido Spanish Hapete haliidid Estonian Azilo-haluro Basque آسیل هالید Persian Savhalogenidek Hungarian Թթուների հալոգենահիդրիտներ Armenian 酸ハロゲン化物 Japanese
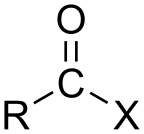
In organic chemistry, an acyl halide (also known as an acid halide) is a chemical compound derived from an oxoacid[1] by replacing a hydroxyl group (−OH) with a halide group (−X, where X is a halogen).[2]
If the acid is a carboxylic acid (−C(=O)OH), the compound contains a −C(=O)X functional group, which consists of a carbonyl group (C=O) singly bonded to a halogen atom.[3] The general formula for such an acyl halide can be written RCOX, where R may be, for example, an alkyl group, CO is the carbonyl group, and X represents the halide, such as chloride. Acyl chlorides are the most commonly encountered acyl halides, but acetyl iodide is the one produced (transiently) on the largest scale. Billions of kilograms are generated annually in the production of acetic acid.[4]
- ^ IUPAC, Compendium of Chemical Terminology, 2nd ed. (the "Gold Book") (1997). Online corrected version: (2006–) "acyl groups". doi:10.1351/goldbook.A00123
- ^ IUPAC, Compendium of Chemical Terminology, 2nd ed. (the "Gold Book") (1997). Online corrected version: (2006–) "acyl halides". doi:10.1351/goldbook.A00124
- ^ Saul Patai, ed. (1972). Acyl Halides. PATAI'S Chemistry of Functional Groups. doi:10.1002/9780470771273. ISBN 9780470771273.
- ^ Hosea Cheung, Robin S. Tanke, G. Paul Torrence "Acetic Acid" in Ullmann's Encyclopedia of Industrial Chemistry 2002, Wiley-VCH, Weinheim. doi:10.1002/14356007.a01_045