Back ثلاثي كلوريد البورون Arabic تریکولورید بور AZB Chlorid boritý Czech Bortrichlorid German بور تریکلرید Persian Booritrikloridi Finnish Trichlorure de bore French बोरोन ट्राइक्लोराइड Hindi Boron triklorida ID Tricloruro di boro Italian
| |||
| Names | |||
|---|---|---|---|
| IUPAC name
Boron trichloride
| |||
| Other names
Boron(III) chloride
Trichloroborane | |||
| Identifiers | |||
3D model (JSmol)
|
|||
| ChemSpider | |||
| ECHA InfoCard | 100.030.586 | ||
| EC Number |
| ||
PubChem CID
|
|||
| RTECS number |
| ||
| UNII | |||
CompTox Dashboard (EPA)
|
|||
| |||
| |||
| Properties | |||
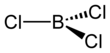
| BCl3 | |||
| Molar mass | 117.17 g/mol | ||
| Appearance | Colorless gas, fumes in air | ||
| Density | 1.326 g/cm3 | ||
| Melting point | −107.3 °C (−161.1 °F; 165.8 K) | ||
| Boiling point | 12.6 °C (54.7 °F; 285.8 K)[1] | ||
| hydrolysis | |||
| Solubility | soluble in CCl4, ethanol | ||
| -59.9·10−6 cm3/mol | |||
Refractive index (nD)
|
1.00139 | ||
| Structure | |||
| Trigonal planar (D3h) | |||
| zero | |||
| Thermochemistry | |||
Heat capacity (C)
|
107 J/mol K | ||
Std molar
entropy (S⦵298) |
206 J/mol K | ||
Std enthalpy of
formation (ΔfH⦵298) |
-427 kJ/mol | ||
Gibbs free energy (ΔfG⦵)
|
-387.2 kJ/mol | ||
| Hazards[2] | |||
| Occupational safety and health (OHS/OSH): | |||
Main hazards
|
May be fatal if swallowed or if inhaled Causes serious burns to eyes, skin, mouth, lungs, etc. Contact with water gives HCl | ||
| GHS labelling: | |||
  
| |||
| Danger | |||
| H300, H314, H330[note 1] | |||
| NFPA 704 (fire diamond) | |||
| Flash point | Non-flammable | ||
| Safety data sheet (SDS) | ICSC | ||
| Related compounds | |||
Other anions
|
Boron trifluoride Boron tribromide Boron triiodide | ||
Other cations
|
Aluminium trichloride Gallium trichloride | ||
Related compounds
|
Boron trioxide Carbon tetrachloride | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||
Boron trichloride is the inorganic compound with the formula BCl3. This colorless gas is a reagent in organic synthesis. It is highly reactive towards water.
- ^ Cite error: The named reference
EROSwas invoked but never defined (see the help page). - ^ Index no. 005-002-00-5 of Annex VI, Part 3, to Regulation (EC) No 1272/2008 of the European Parliament and of the Council of 16 December 2008 on classification, labelling and packaging of substances and mixtures, amending and repealing Directives 67/548/EEC and 1999/45/EC, and amending Regulation (EC) No 1907/2006. OJEU L353, 31.12.2008, pp 1–1355 at p 341.
Cite error: There are <ref group=note> tags on this page, but the references will not show without a {{reflist|group=note}} template (see the help page).