Back تأثير ليدنفروست Arabic Эфект Лейдэнфроста Byelorussian Efecte Leidenfrost Catalan Leidenfrostův jev Czech Leidenfrost-effekten Danish Leidenfrost-Effekt German Φαινόμενο Λάιντενφροστ Greek Leidenfrost-efiko Esperanto Efecto Leidenfrost Spanish پدیده لایدنفروست Persian


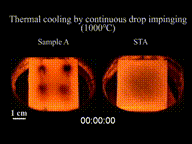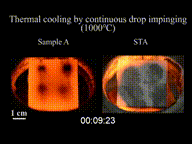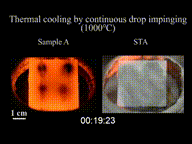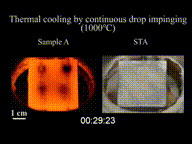
The Leidenfrost effect is a physical phenomenon in which a liquid, close to a solid surface of another body that is significantly hotter than the liquid's boiling point, produces an insulating vapor layer that keeps the liquid from boiling rapidly. Because of this repulsive force, a droplet hovers over the surface, rather than making physical contact with it. The effect is named after the German doctor Johann Gottlob Leidenfrost, who described it in A Tract About Some Qualities of Common Water.
This is most commonly seen when cooking, when drops of water are sprinkled onto a hot pan. If the pan's temperature is at or above the Leidenfrost point, which is approximately 193 °C (379 °F) for water, the water skitters across the pan and takes longer to evaporate than it would take if the water droplets had been sprinkled onto a cooler pan.