
Back مغنيسيوم في علم الأحياء Arabic Papel biológico del magnesio Spanish Magnesioa biologian Basque منیزیم در زیستشناسی Persian आहारीय मैग्नेशियम Hindi Magnesium dalam biologi ID Biyolojide magnezyum Turkish Магній у біології Ukrainian Magnesi trong sinh học Vietnamese
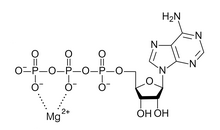
Magnesium is an essential element in biological systems. Magnesium occurs typically as the Mg2+ ion. It is an essential mineral nutrient (i.e., element) for life[1][2][3][4] and is present in every cell type in every organism. For example, adenosine triphosphate (ATP), the main source of energy in cells, must bind to a magnesium ion in order to be biologically active. What is called ATP is often actually Mg-ATP.[5] As such, magnesium plays a role in the stability of all polyphosphate compounds in the cells, including those associated with the synthesis of DNA and RNA.[citation needed]
Over 300 enzymes require the presence of magnesium ions for their catalytic action, including all enzymes utilizing or synthesizing ATP, or those that use other nucleotides to synthesize DNA and RNA.[6]
In plants, magnesium is necessary for synthesis of chlorophyll and photosynthesis.[citation needed]
- ^ "Magnesium (In Biological Systems)". Van Nostrand's Scientific Encyclopedia. John Wiley & Sons, Inc. 2006. doi:10.1002/0471743984.vse4741. ISBN 978-0471743989.
- ^ Leroy J (1926). "Necessite du magnesium pour la croissance de la souris". Comptes Rendus des Séances de la Société de Biologie. 94: 431–433.
- ^ Lusk JE, Williams RJ, Kennedy EP (1968). "Magnesium and the growth of Escherichia coli". Journal of Biological Chemistry. 243 (10): 2618–2624. doi:10.1016/S0021-9258(18)93417-4. PMID 4968384.
- ^ Marschner H (1995). Mineral Nutrition in Higher Plants. San Diego: Academic Press. ISBN 978-0-12-473542-2.
- ^ "Definition: magnesium from Online Medical Dictionary". 25 December 2007. Archived from the original on 25 December 2007. Retrieved 17 January 2018.
- ^ Dietary Reference Intakes for Calcium, Phosphorus, Magnesium, Vitamin D, and Fluoride. Washington, DC: National Academy Press. 1997. pp. 190–191. doi:10.17226/5776. ISBN 978-0-309-06403-3. PMID 23115811.