 | |
| Clinical data | |
|---|---|
| Trade names | Colprone, others |
| Other names | Metrogestone; Medrogesterone; AY-62022, NSC-123018, R-13615; 6,17α-Dimethyl-6-dehydroprogesterone; 6,17α-Dimethyl-4,6-pregnadiene-3,20-dione |
| Pregnancy category |
|
| Routes of administration | By mouth |
| Drug class | Progestogen; Progestin |
| ATC code | |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Bioavailability | Nearly 100%[1][2] |
| Protein binding | 95%: Albumin (90%), CBG (3%), SHBG (2%)[2] |
| Metabolism | Hepatic (hydroxylation)[1] |
| Elimination half-life | 35–36 hours[3][4][5] |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.012.323 |
| Chemical and physical data | |
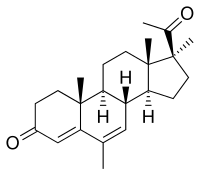
| Formula | C23H32O2 |
| Molar mass | 340.507 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
Medrogestone, sold under the brand name Colprone among others, is a progestin medication which has been used in menopausal hormone therapy and in the treatment of gynecological disorders.[6][2] It is available both alone and in combination with an estrogen.[7] It is taken by mouth.[2][8]
Medrogestone is a progestin, or a synthetic progestogen, and hence is an agonist of the progesterone receptor, the biological target of progestogens like progesterone.[2] It has weak antiandrogenic, glucocorticoid, and antimineralocorticoid activity and no other important hormonal activity.[2][1][9][10] Due to its progestogenic activity, medrogestone has antigonadotropic effects.[1][2]
Medrogestone was described as early as 1963 and was introduced for medical use by at least 1966.[11][12][9] It has mostly been discontinued and remains available only in a few countries.[13][7]
- ^ a b c d Schindler AE, Campagnoli C, Druckmann R, Huber J, Pasqualini JR, Schweppe KW, Thijssen JH (December 2003). "Classification and pharmacology of progestins" (PDF). Maturitas. 46 (Suppl 1): S7–S16. doi:10.1016/j.maturitas.2003.09.014. PMID 14670641.[permanent dead link]
- ^ a b c d e f g Kuhl H (2005). "Pharmacology of estrogens and progestogens: influence of different routes of administration" (PDF). Climacteric. 8 (Suppl 1): 3–63. doi:10.1080/13697130500148875. PMID 16112947. S2CID 24616324.
- ^ Stanczyk FZ (2002). "Pharmacokinetics and potency of progestins used for hormone replacement therapy and contraception". Reviews in Endocrine & Metabolic Disorders. 3 (3): 211–24. doi:10.1023/A:1020072325818. PMID 12215716. S2CID 27018468.
Medrogestone The pharmacokinetics of medrogestone (5 mg dose) was studied in 12 Chinese young males who received a single oral dose of this drug [20]. The mean ± standard deviation Cmax was 8.21 ± 2.78 ng/ml and Tmax was 2.57 ± 1.02; the half-life of elimination was 34.9 ± 17.0 hours.
- ^ Lobo R, Crosignani P, Paoletti R, Bruschi F (6 December 2012). Women's Health and Menopause: New Strategies — Improved Quality of Life. Springer Science & Business Media. pp. 142–. ISBN 978-1-4615-1061-1.
- ^ Stanczyk FZ, Hapgood JP, Winer S, Mishell DR (April 2013). "Progestogens used in postmenopausal hormone therapy: differences in their pharmacological properties, intracellular actions, and clinical effects". Endocrine Reviews. 34 (2): 171–208. doi:10.1210/er.2012-1008. PMC 3610676. PMID 23238854.
- ^ Cite error: The named reference
MortonHall1999was invoked but never defined (see the help page). - ^ a b Cite error: The named reference
Drugs.comwas invoked but never defined (see the help page). - ^ Cutler WB, Garcia CR (1993). Menopause: A Guide for Women and Those who Love Them. Norton. pp. 134–. ISBN 978-0-393-30995-9.
- ^ a b Cite error: The named reference
ReveszChappel1966was invoked but never defined (see the help page). - ^ Cite error: The named reference
pmid4105445was invoked but never defined (see the help page). - ^ Cite error: The named reference
Elks2014was invoked but never defined (see the help page). - ^ Cite error: The named reference
DeghenghiLefebvre1963was invoked but never defined (see the help page). - ^ Cite error: The named reference
IndexNominum2000was invoked but never defined (see the help page).
