
Back بيروكسي أحادي كبريتات البوتاسيوم Arabic پوتاسیوم پروکسیومونوسولفات AZB Hydrogenperoxosíran draselný Czech Kaliumperoxomonosulfat German Peroximonosulfato de potasio Spanish پتاسیم پروکسیمونوسولفات Persian Kaliumperoksimonosulfaatti Finnish Hydrogénopersulfate de potassium French पोटासियम परऑक्सीमोनोसल्फेट Hindi ペルオキシ一硫酸カリウム Japanese
| |
| Names | |
|---|---|
| IUPAC name
Potassium peroxysulfate
| |
| Other names | |
| Identifiers | |
| |
3D model (JSmol)
|
|
| ChemSpider | |
| ECHA InfoCard | 100.030.158 |
PubChem CID
|
|
| UNII |
|
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
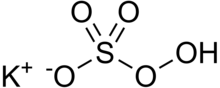
| KHSO5 | |
| Molar mass | 152.2 g/mol (614.76 g/mol as triple salt) |
| Appearance | Off-white powder |
| Decomposes | |
| Hazards | |
| Occupational safety and health (OHS/OSH): | |
Main hazards
|
Oxidant, corrosive |
| NFPA 704 (fire diamond) | |
| Safety data sheet (SDS) | Degussa Caroat MSDS |
| Related compounds | |
Related compounds
|
Potassium persulfate |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Potassium peroxymonosulfate is widely used as an oxidizing agent, for example, in pools and spas (usually referred to as monopersulfate or "MPS"). It is the potassium salt of peroxymonosulfuric acid. Usually potassium peroxymonosulfate is available as the triple salt 2KHSO5·KHSO4·K2SO4, known as Oxone.
The standard electrode potential for potassium peroxymonosulfate is +1.81 V with a half reaction generating the hydrogen sulfate (pH = 0):[4]
- HSO−5 + 2H+ + 2e− → HSO−4 + H2O
- ^ Wu, Mingsong; Xu, Xinyang; Xu, Xun (November 2014). "Algicidal and Bactericidal Effect of Potassium Monopersulfate Compound on Eutrophic Water". Applied Mechanics and Materials. 707: 259. doi:10.4028/www.scientific.net/AMM.707.259. S2CID 98000605.
- ^ Pool School. Trouble Free Pool. p. PT4. Retrieved November 30, 2018.
- ^ "DuPont MSDS" (PDF). Archived from the original (PDF) on 2014-08-15. Retrieved 2012-01-26.
- ^ Spiro, M. (1979). "The standard potential of the peroxosulphate/sulphate couple". Electrochimica Acta. 24 (3): 313–314. doi:10.1016/0013-4686(79)85051-3. ISSN 0013-4686.
