Back حمض ثلاثي كلورو الخليك Arabic تری کولورواستیک اسید AZB Kyselina trichloroctová Czech Trikloreddikesyre Danish Trichloressigsäure German Trikloroacetata acido Esperanto Ácido tricloroacético Spanish Azido trikloroazetiko Basque تری کلرواستیک اسید Persian Trikloorietikkahappo Finnish
| |||

| |||
| Names | |||
|---|---|---|---|
| Preferred IUPAC name
Trichloroacetic acid | |||
| Identifiers | |||
3D model (JSmol)
|
|||
| 970119 | |||
| ChEBI | |||
| ChEMBL | |||
| ChemSpider | |||
| ECHA InfoCard | 100.000.844 | ||
| 2842 | |||
| KEGG | |||
PubChem CID
|
|||
| RTECS number |
| ||
| UNII | |||
CompTox Dashboard (EPA)
|
|||
| |||
| |||
| Properties | |||
| C2HCl3O2 | |||
| Molar mass | 163.38 g·mol−1 | ||
| Appearance | Colorless to white, crystalline solid | ||
| Odor | Sharp, pungent [1] | ||
| Density | 1.63 g/cm3 | ||
| Melting point | 57 to 58 °C (135 to 136 °F; 330 to 331 K)[2] | ||
| Boiling point | 196 to 197 °C (385 to 387 °F; 469 to 470 K)[2] | ||
| 1000 g/100 mL[2] | |||
| Vapor pressure | 1 mmHg (51.1 °C)[1] | ||
| Acidity (pKa) | 0.66[3] | ||
| −73.0·10−6 cm3/mol | |||
| Structure | |||
| 3.23 D | |||
| Hazards | |||
| GHS labelling: | |||
 
| |||
| Danger | |||
| H314, H410 | |||
| P260, P264, P273, P280, P301+P330+P331, P303+P361+P353, P304+P340, P305+P351+P338, P310, P321, P363, P391, P405, P501 | |||
| NFPA 704 (fire diamond) | |||
| Lethal dose or concentration (LD, LC): | |||
LD50 (median dose)
|
5000 mg/kg orally in rats[2] | ||
| NIOSH (US health exposure limits): | |||
PEL (Permissible)
|
None[1] | ||
REL (Recommended)
|
TWA 1 ppm (7 mg/m3)[1] | ||
IDLH (Immediate danger)
|
N.D.[1] | ||
| Related compounds | |||
Related chloroacetic acids
|
Chloroacetic acid Dichloroacetic acid | ||
Related compounds
|
Acetic acid Trifluoroacetic acid Tribromoacetic acid | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||
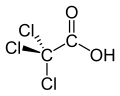
Trichloroacetic acid (TCA; TCAA; also known as trichloroethanoic acid) is an analogue of acetic acid in which the three hydrogen atoms of the methyl group have all been replaced by chlorine atoms. Salts and esters of trichloroacetic acid are called trichloroacetates.
- ^ a b c d e NIOSH Pocket Guide to Chemical Hazards #0626 National Institute for Occupational Safety and Health (NIOSH)
- ^ a b c d Budavari, Susan, ed. (1996), The Merck Index: An Encyclopedia of Chemicals, Drugs, and Biologicals (12th ed.), Merck, ISBN 0911910123
- ^ Databog fysik kemi, F&K Forlaget 11. udgave 2009