
Back Uraan(VI)fluoried Afrikaans سداسي فلوريد اليورانيوم Arabic هقزافلوراید اورانیوم AZB Hexafluorur d'urani Catalan Fluorid uranový Czech Uran(VI)-fluorid German Hexafluoruro de uranio Spanish Uranio hexafluoruro Basque هگزافلوراید اورانیوم Persian Uraaniheksafluoridi Finnish
You can help expand this article with text translated from the corresponding article in German. (November 2012) Click [show] for important translation instructions.
|
| |
| |
| |
| Names | |
|---|---|
| IUPAC names
Uranium hexafluoride
Uranium(VI) fluoride | |
| Identifiers | |
3D model (JSmol)
|
|
| Abbreviations | hex |
| ChEBI | |
| ChemSpider | |
| ECHA InfoCard | 100.029.116 |
| EC Number |
|
| 2923 | |
PubChem CID
|
|
| RTECS number |
|
| UNII | |
| UN number | 2978 (<1% 235U) 2977 (>1% 235U) |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| UF6 | |
| Molar mass | 352.02 g/mol |
| Appearance | Colorless solid |
| Density | 5.09 g/cm3, solid |
| Boiling point | 56.5 °C (133.7 °F; 329.6 K) (sublimes, at atmospheric pressure) |
| Hydrolyzes | |
| Solubility |
|
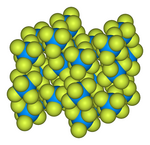
| Structure | |
| Orthorhombic, oP28 | |
| Pnma, No. 62 | |
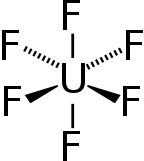
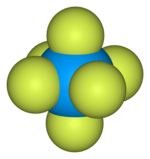
| Octahedral (Oh) | |
| 0 | |
| Thermochemistry | |
Std molar
entropy (S⦵298) |
|
Std enthalpy of
formation (ΔfH⦵298) |
|
| Hazards | |
| Occupational safety and health (OHS/OSH): | |
Main hazards
|
Toxic, corrosive, radioactive[3] |
| GHS labelling: | |
  
| |
| Danger | |
| H300, H330, H373, H411 | |
| NFPA 704 (fire diamond) | |
| Flash point | Non-flammable |
| Safety data sheet (SDS) | ICSC 1250 |
| Related compounds | |
Other anions
|
Uranium hexachloride |
Other cations
|
|
Related uranium fluorides
|
|
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Uranium hexafluoride, sometimes called hex, is an inorganic compound with the formula UF6. Uranium hexafluoride is a volatile and toxic white solid that reacts with water, releasing corrosive hydrofluoric acid. The compound reacts mildly with aluminium, forming a thin surface layer of AlF3 that resists any further reaction from the compound. UF6 is used in the process of enriching uranium, which produces fuel for nuclear reactors and nuclear weapons.
- ^ "Uranium Hexafluoride". Archived from the original on 2013-09-16. Retrieved 2013-08-08.
- ^ a b c d Johnson, Gerald K. (1979). "The Enthalpy of Formation of Uranium Hexafluoride". The Journal of Chemical Thermodynamics. 11 (5): 483–490. doi:10.1016/0021-9614(79)90126-5.
- ^ Uranium(VI) fluoride
